Dekoracja ściany z klocków drewnianych ciętych w poprzek włókien

Do naturalnych materiałów dekoracyjnych, stosowanych wewnątrz i na zewnątrz pomieszczeń, należy bezsprzecznie kamień. Z reguły wykłada się nim kominki, małe fragmenty ścian lub filary. Również zieleń w pokojowym miniogródku obmurowanym kamieniem i ewentualnie uzupełnionym fontanną, ze stale tryskającą wodą może być uroczym zakątkiem mieszkania.

Kontynuacją kamiennego ogródka w kierunku stropu może być dekoracja z drewna, której kształt i sposób ułożenia będzie podobny do kamiennego murku. Licową powierzchnię dekoracji tworzyć będą elementy z drewna ciętego w poprzek włókien, którego słoje stanowić będą szereg centrycznych pierścieni. Do tych celów najlepiej nadawać się będzie drewno wyraziście słoiste, np. modrzew, sosna lub świerk.
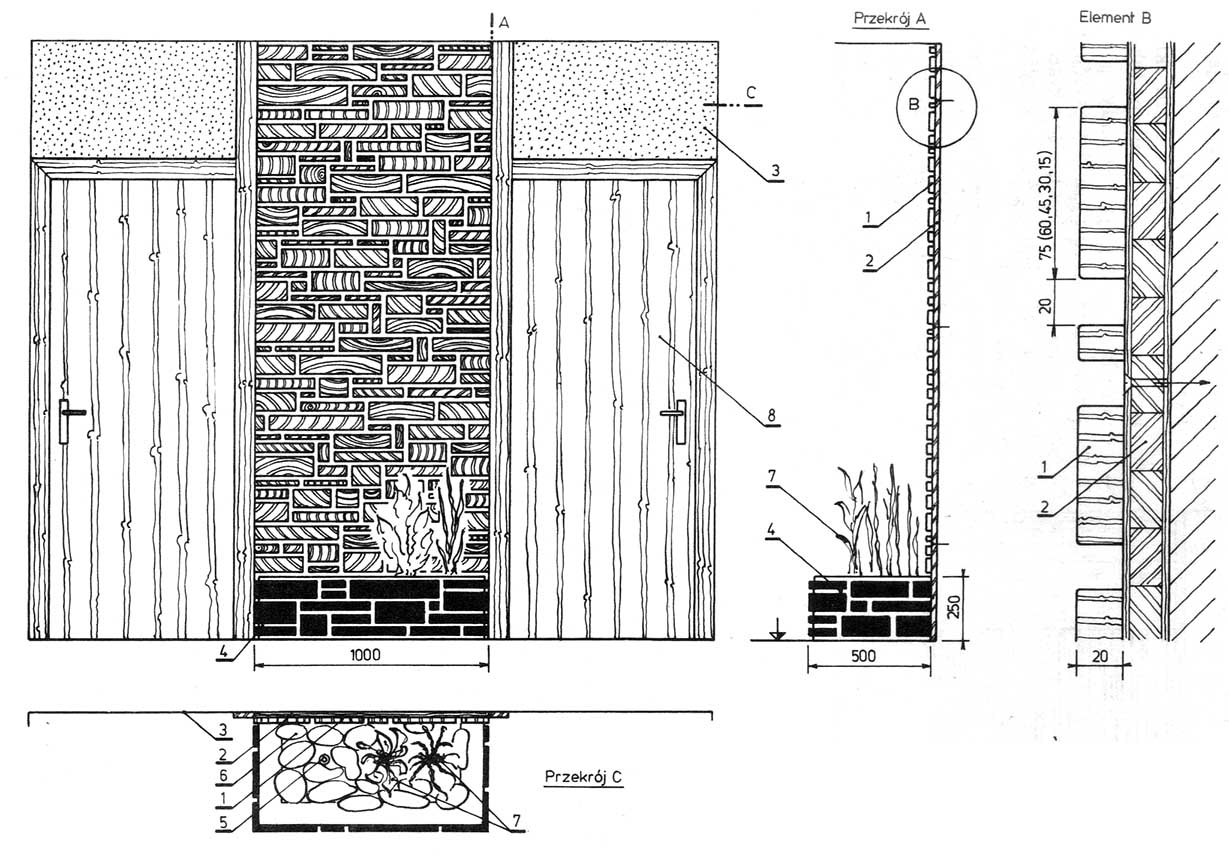
Rysunek. Kompozycja dekoracji ścianki wyłożonej klockami z drzwiami i tynkiem ozdobnym: 1 — klocki cięte poprzecznie, 2 — płyta stolarska, 3 — tynk ozdobny, 4 — ogródek z kamiennego muru, 5 — nawilżacz powietrza (mini fontanna), 6 — kamienie, 7 — rośliny pokojowe, 8 — drzwi w naturalnej okleinie.
Właściwym uzupełnieniem tej kombinacji materiałów może być szlachetny tynk nad drzwiami i kasetonowy strop z drewna klonu. Alternatywne rozwiązanie zniżonej do wysokości drzwi wykładziny i szlachetnego tynku pokazuje rysunku.
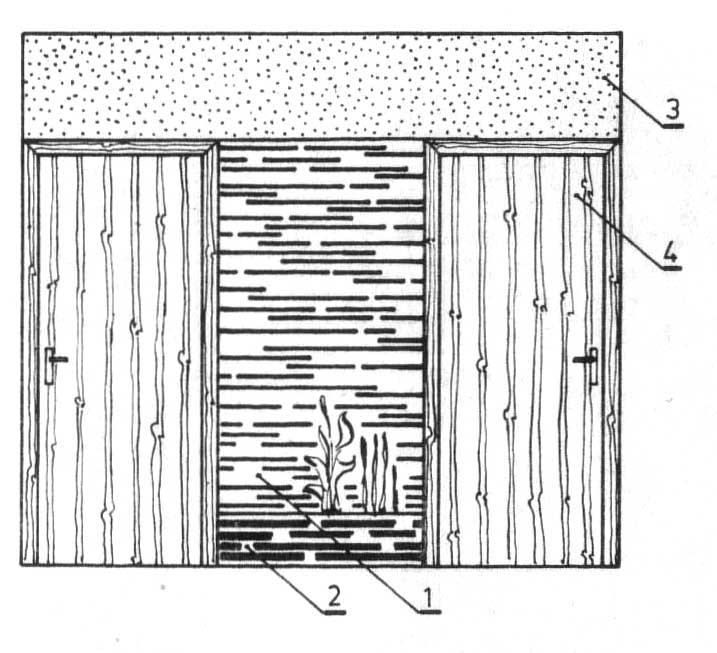
Rysunek. Dekoracja z klocków do wysokości drzwi; rozwiązanie alternatywne do rys powyżej: 1 — dekoracja z klocków, 2 — ogródek z kamiennego muru, 3 — tynk ozdobny, 4 — drzwi w naturalnej okleinie.
Produkcja klocków jest stosunkowo prosta, z wyjątkiem obróbki wykończającej dużej liczby elementów.

Dobrze wysuszoną tarcicę obrzynaną, o dowolnej długości, różnej grubości i szerokości 5—20 cm, struga się czterostronnie na grubościówce. Po przestruganiu powinno się otrzymać w przybliżeniu taką samą ilość materiału w pięciu klasach grubości, tj. 15, 30, 45, 60 i 75 mm. Następnie szlifuje się przestrugane płaszczyzny celem uniknięcia szlifowania małych klocków.
Uzyskane w ten sposób fryzy przerzyna się na pilarce tarczowej na klocki długości 20 mm. Na szlifierce tarczowej obrabia się jedną płaszczyznę czołową każdego klocka, a papierem ściernym zaokrągla osiem widocznych krawędzi.
Obrobione klocki przykleja się do przyciętej na odpowiedni wymiar i przeszlifowanej płyty stolarskiej. Otwory montażowe w płycie wykonuje się po naklejeniu klocków, w szczelinach między klockami. Do przyklejania klocków drewnianych należy użyć kleju dającego elastyczne złącze (np. butaprenu). Klej nanosi się na klocek punktowo, aby się on nie deformował. Sposób nakładania i rodzaj kleju jest podyktowany zmianami wilgotności powietrza w okresie letnim i zimowym oraz znaczną szerokością klocków (do 20 cm) powodującymi okresowe zmiany jego wymiarów dochodzące do 0,5 mm. Brak elastycznego lepiszcza spowoduje popękanie klocków.
Wybór kompozycji układu klocków w wykładzinie muropodobnej określają następujące zasady:
— szczeliny (fugi) między klockami mogą tworzyć jedynie układ litery T; nigdy układ krzyżowy (rysunek);
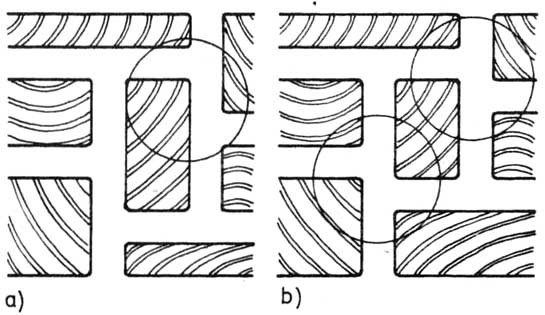
Rysunek. Przemienność szczelin (przerw) między klockami: a) prawidłowa (szczelina w kształcie litery T), b) nieprawidłowa (szczelina krzyżowa).
— szczeliny (fugi) poziome powinny być przesunięte względem siebie o co najmniej jedną szerokość najwęższego klocka (rysunek).
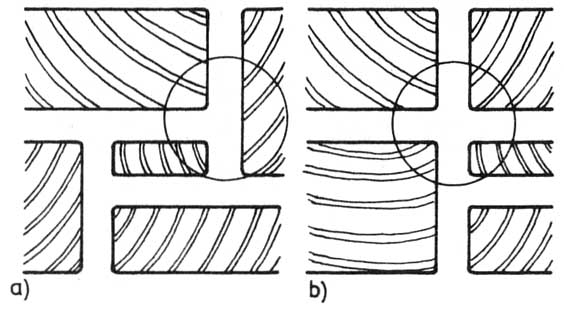
Rysunek. Pozioma przemienność szczelin: a) prawidłowa (przemienność przez jedną grubość), b) nieprawidłowy splot szczelin poziomych.
Podobne zasady powinny obowiązywać również przy wykonywaniu kamiennego muru.
Po naklejeniu wszystkich klocków i wyschnięciu kleju całą płytę stolarską z wykładziną klockową kilkakrotnie lakieruje się metodą natrysku. Po każdej warstwie lakieru przeszlifowuje się jedynie płaszczyzny czołowe klocków. Pył po szlifowaniu należy dokładnie wydmuchać ze szczelin. W wyniku kilkukrotnego natrysku lakieru wypełnione zostaną wszystkie pory w płaszczyźnie czołowej klocków i powstanie jednolita, gładka powierzchnia. Lakierować można na mat lub połysk. Po wyschnięciu lakieru płytę z wykładziną przykręca się do ściany.
